The IBC is federally mandated in the NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Modules. The IBC reviews research involving biohazards and inspects/certifies labs using the CDC Biosafety in Microbiological and Biomedical Labs (BMBL). The IBC’s main goal is the protection of public health, environmental protection, and preventing economic harm from biological agents.
An IBC is comprised of at least 5 members containing: 1 animal expert, 1 plant expert, 1 non-scientist, 2 local unaffiliated persons.
Functions
- Review protocol registrations for research and teaching activities involving biological agents.
- Review or develop Standard Operating Procedures (SOPs) for research and teaching activities involving biological agents and safety in biosafety, plant biosafety, and animal biosafety labs.
- Inspect and certify labs where biological agents are used.
- Review and investigate legitimate concerns in research projects involving biohazards and biological agents.
- Make recommendations to the Institutional Official regarding any aspect of the research facility’s biosafety program, facilities, or personnel training.
- Suspend an activity involving biological agents when necessary; take corrective action and report to the funding agency
What May Fall Under the IBC
- Recombinant DNA
- Synthetic Nucleic Acid Modules
- Human pathogens
- Animal diseases
- Viral agents
- Vectors
- Human biological material
- Bacteria
- Plant diseases
- Inoculants
Submission Process
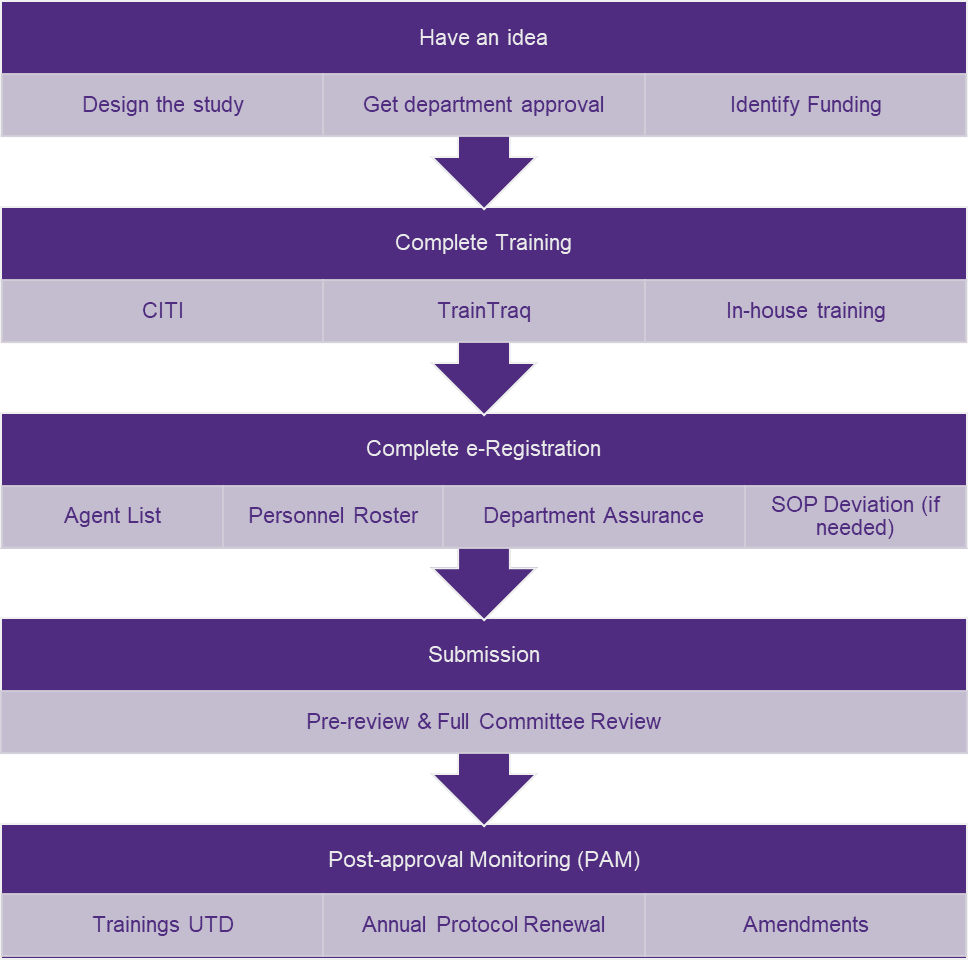
- The use biological agents falls under a continuing review
- Layman’s descriptions should be simple so any non-scientist can understand.
- Agents and strains should be spelled correctly and consistent
- Expires every 3 years
| Citi | Traintraq |
|---|---|
| Introduction to Biosafety | Bloodborne Pathogens for Research Personnel |
| Biosafety Refresher | Lab Safety |
| Introduction to Research Compliance Requirements | |
| Biosafety Level 1 or 2 |
*Other trainings may be required based on project requirements
*Researchers must complete annual occupational health and safety enrollment
The IBC convenes on the third Tuesday of each month, with adjustments made when holidays require it. Protocols must be submitted at least four weeks prior to the third Tuesday of the month in order to be reviewed at that meeting. All meetings are held virtually.
Be sure to plan ahead if your protocol also requires approval from other compliance programs, such as the IACUC or IRB.
Details of the meeting dates and deadlines can be found here
Contact Information
Contact the IBC at: ibc@tarleton.edu | phone 254-459-5563 or 254-576-6030